Our planet has a plastic problem. Although plastic is incredibly useful, our current way of dealing with it has frightening repercussions. Can we find a better way of doing things?
An estimated 359 million tonnes of plastics were produced in 2018 alone, so what can we do about it? The answer is found in the famous phrase: reduce, reuse, recycle. We can reduce plastic consumption by designing intelligently, considering plastic alternatives or biodegradable plastics where it makes sense. Reuse is also part of the equation, with sturdier plastic options–like bags for life–replacing their single-use cousins.
What about plastic that serves a vital function but has reached the end of its useful life? What do you do with a syringe that has delivered vital medicine, or a cracked computer case? We need an effective method to divert this waste plastic from the landfill and transform it into something useful again. Enter: recycling.
The problem with plastic recycling
Our current problem with plastic recycling is that we aren’t doing it enough. Between 1950 and 2015, only 9% of the total plastic produced around the world was recycled, whilst nearly 60% of virgin plastics were used once and discarded. The figure below visualises what this plastics system looks like, with the tiny green section at the bottom showing reuse and recycling.
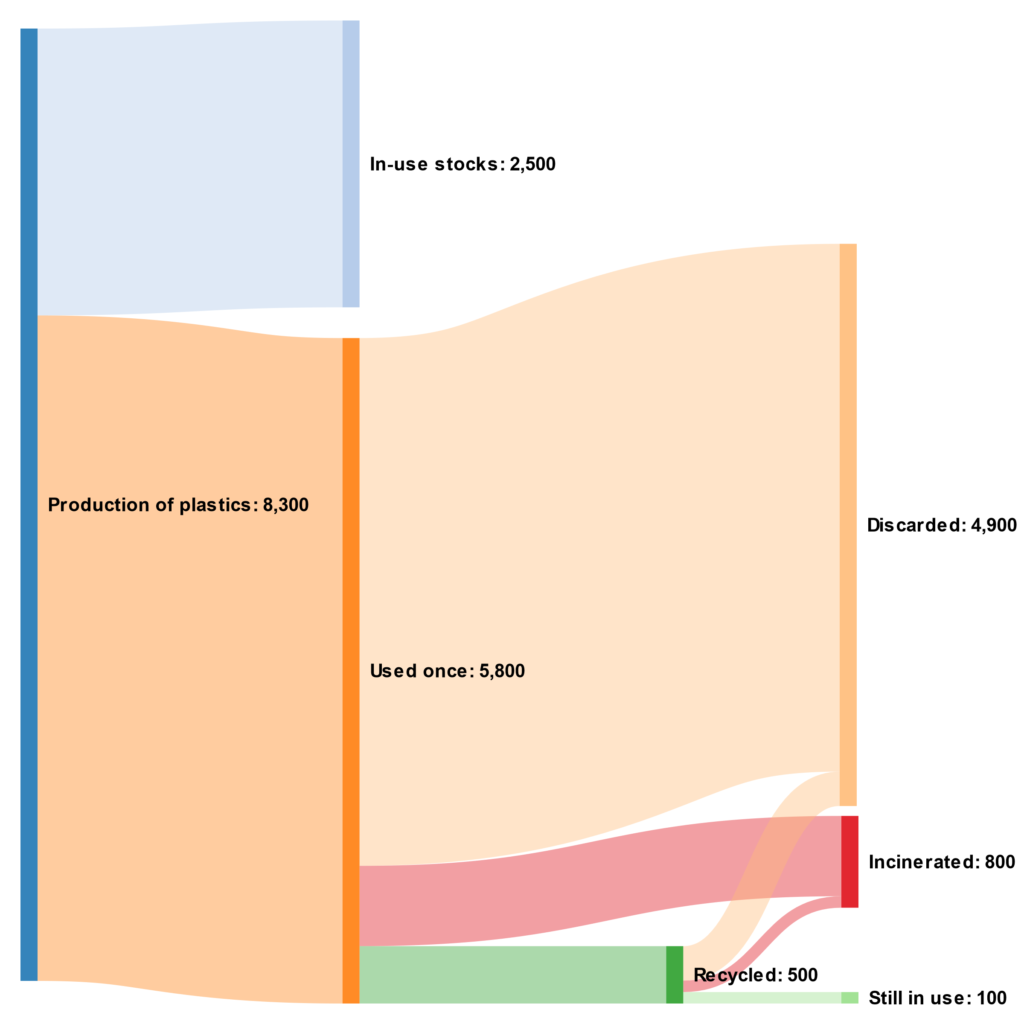
Global plastic production from 1950 to 2015; in million metric tons. Data from Science.
Clearly, we need to recycle more plastic. Most of the plastic recycling that’s done today is mechanical recycling. This process begins with the identification and sorting of plastic waste, which is both time-consuming and expensive–the average sorting machine costs over £250,000. The small pool of suitable plastics is then melted and remoulded into what are essentially poorer quality plastic products. Each time plastic goes through this process it becomes weaker and more brittle, gradually degrading until it needs to be discarded or incinerated.
Chemical recycling of plastics
Chemical recycling is an alternative approach that deconstructs plastics at a more fundamental level. The term covers a variety of processes, each breaking down plastic in a different way.
Plastics are generally derived from crude oil, natural gas or coal and are made up of small building blocks called monomers (mono = one). These monomers are chemically bonded to form long tangled chains of mostly carbon and hydrogen atoms called polymer (poly = many) chains.
In chemical recycling, the polymer chains that form the structure of plastics are manipulated via three different processes: solvent purification, chemical depolymerisation, or thermal depolymerisation. We’ve explained each process below, and compared them to equivalent methods of clothing recycling.
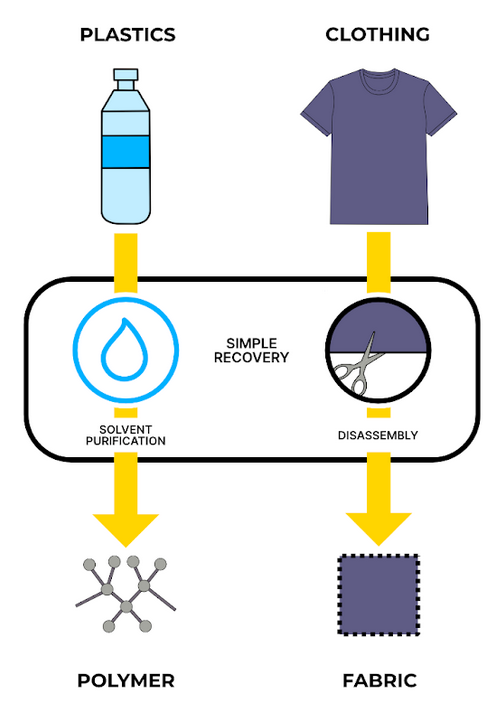
Solvent purification
Here, solvents are used to break down the plastic without changing its chemical structure. The plastic is shredded, and a selective solvent dissolves the plastic, untangling and separating the polymer chains. Any contaminants, like pigments and plasticisers, are removed. The polymer is then re-solidified and treated, resulting in a purified, high-quality plastic.
Solvent-purified plastic is not quite as fresh as virgin plastic, as the polymer chains are degraded slightly during the manufacturing process, but the quality is higher than mechanically recycled plastic. However, solvent recycling requires homogeneous input of only one type of plastic, which is hard to get from post-consumer sources. On the plus side, this method can separate the polyester and cotton components that constitute polycotton blends, which is hard to do by other means.
The graphic below illustrates that recycling plastics via solvent purification is like recycling clothing by disassembling it into squares of fabric. The fabric is cleaned and then stitched into another product–but you’ll lose a bit of fabric at the edges, and to efficiently reuse it you’ll need a big pile of clothing made of the same material.
| Pros | Cons |
|---|---|
| Can separate polycotton textile blends | Only handles homogenous inputs |
| Environmentally friendly solvents can be used | Extensive pre-sorting and pre-treatment steops can be required |
| Solvent can be recovered for reuse | High energy requirements |
| Can recover valuable by-products | Cannot usually remove all contaminants |
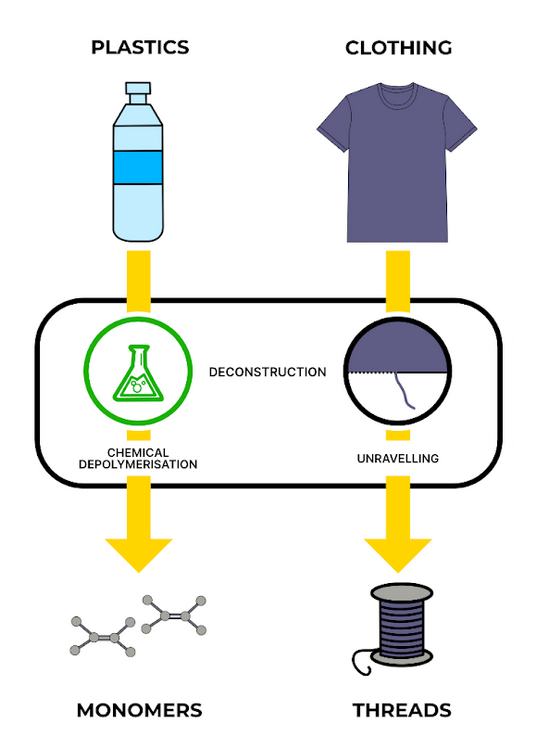
Chemical depolymerisation
Depolymerisation means the polymer chain is broken down into monomers or oligomers (polymer fragments), in this case through a chemical process. The fragments are recovered and purified by separating them from contaminants, and can then be used to build new polymer chains. The purified monomers are identical to those used to create virgin plastics, so new plastics made by this method are top quality. However, depolymerisation can only be done with specific types of plastics, and again usually requires a homogenous input.
In the clothing analogy, depolymerisation is like unravelling the clothing into undyed threads which can be used to weave an entirely new fabric. You get a higher-quality material with more options for the end product, but only if you’re careful not to mix your cotton with your acrylic.
| Pros | Cons |
|---|---|
| Polymers are indistinguishable from virgin plastics | Can only be applied to ‘condensation’ polymers |
| Catalysts and solvents can be recovered | Often requires high energy inputs |
| High yields have been demonstrated | Usually only handles homogenous inputs |
| Commercially viable for bottle and fibre inputs | Lack of information about the process |
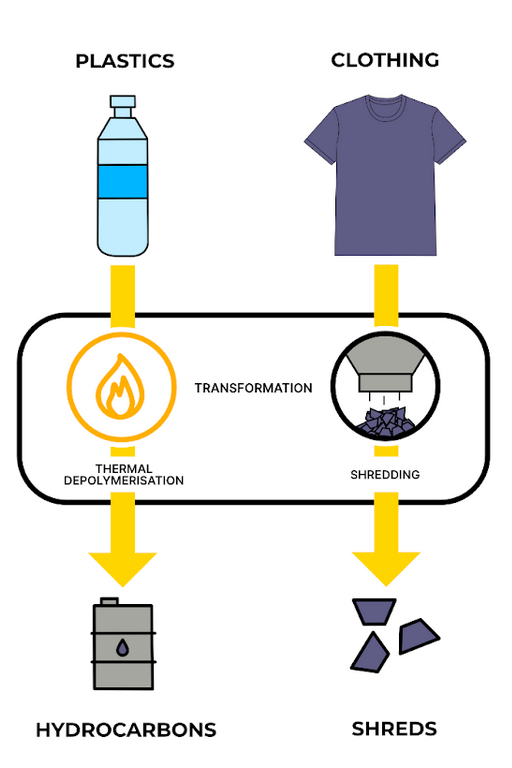
Thermal depolymerisation
Here, polymers are again broken down into small fragments, but this time by applying intense heat. First, the plastic is cleaned, dried, and shredded. Then it’s heated in a controlled reactor under specific conditions to create a mix of chemical fragments. The mixture is purified to obtain the desired product.
The product is usually several steps more basic than a monomer, as the polymer chain is broken down chaotically into fundamental components, and would need extensive processing to make a new polymer. However, this is the only chemical recycling process that can handle mixed waste streams.
There are two main processes:
- Pyrolysis (heating at moderate to high temperature in the absence of oxygen) – produces basic hydrocarbon products including gases, oils, and waxes
- Gasification (heating at very high temperatures in limited oxygen) – produces syngas, which can be burnt for energy or used to produce new hydrocarbons
Thermal depolymerisation is like ripping a t-shirt into shreds of fabric that couldn’t easily be used to make a new t-shirt, but could be used in other ways, like stuffing a pillow. Different fabric types could be mixed together for shredding, with no sorting needed.
| Pros | Cons |
|---|---|
| Can accept more heterogeneous mixtures of waste at the cost of quality | Requires a homogenous source of plastic for better outputs |
| Can be applied to certain non-recyclable waste streams | Complex mixture of products |
| Outputs can manufacture plastics of equal quality to virgin feedstocks | Outputs currently skewed towards producing low-cost fuel |
| High temperatures effectively decontaminate the inputs | No information on the recycling of by-products and reagents |
Chemical recycling outlook
Only 12% of the 260 million tonnes of global plastic waste were recycled in 2016, and plastic waste is set to double by 2030. This is alarming, but chemical recycling has the potential to complement mechanical recycling as part of the plastics solution.
Chemical recycling holds promise for niche applications that mechanical recycling isn’t cut out for, but currently has limited scope due to its comparatively high energy demand and a lack of transparency around the process. More development and incentives are needed to turn chemical recycling into an option that’s usable at a massive scale.
Ultimately, a sustainable future requires a serious effort to minimise fossil fuel consumption and find ways to reduce plastic production.
Green Rose Chemistry uses unbiased science to move industry toward a bright green future. Read more about what we do here.
Further Reading
Plastic pollution: how chemical recycling technology could help fix it
Chemical Recycling: State ofPlay

